 
The overall electrophilic aromatic substitutions are exergonic reactions. It has a lower free energy of activation. The second step is fast and exergonic because it restores the stability enhancing aromaticity. This step is therefore the rate-determining step because of its higher free energy of activation. The free energy diagram shows that the first step is relatively slow and endergonic because the ring loses its aromatic stability. In the second step, deprotonation of the arenium ion restores aromaticity and gives the substituted product. It is often referred to as a sigma complex because the electrophile forms a sigma bond with the aromatic ring. In the first step, the π system of the aromatic ring reacts with an electrophile, forming an arenium ion, which is resonance-stabilized. Introduction Aromatic compounds are especially stable and despite having p-bonds do not react like typical alkenes. All electrophilic aromatic substitution reactions occur via a two-step mechanism. Many functional groups can be added to aromatic compounds by these reactions. In an electrophilic aromatic substitution reaction, an electrophile substitutes for a hydrogen of an aromatic compound. Overall, electrophilic aromatic substitutions are exergonic reactions. The nature of substituents on the aromatic ring influences its reactivity towards electrophilic substitution reactions. It has lower free energy of activation and is fast. For example, nitration in mixtures of nitric and sulfuric acids is not brought about by attack of the nitric. In contrast, the second step is exergonic because it restores aromatic stability to the system. It is important to realize that in aromatic substitution the actual electrophilic substituting agent, X or X Y, is not necessarily the reagent that is added to the reaction mixture. It is, therefore, the rate-determining step. This step has a higher free energy of activation and is slow. In the second step, the arenium ion is deprotonated, restoring aromaticity and giving the substituted product.Īs evident from the free energy diagram, the first step is endergonic because the ring loses its aromatic stability. The arenium ion is also called a sigma complex because the electrophile forms a sigma bond with the aromatic ring. Here, we demonstrate successful EAS reactions of Craig-Möbius aromatics, osmapentalenes, and fused osmapentalenes. In the first step of the reaction mechanism, the π system of the aromatic ring attacks the electrophile to form an arenium ion, which is resonance stabilized. Table 3-2: MPEX, Harel and Hammer Analogies. Electrophilic aromatic substitution (EAS) reactions are widely regarded as characteristic reactions of aromatic species, but no comparable reaction has been reported for molecules with Craig-Möbius aromaticity. 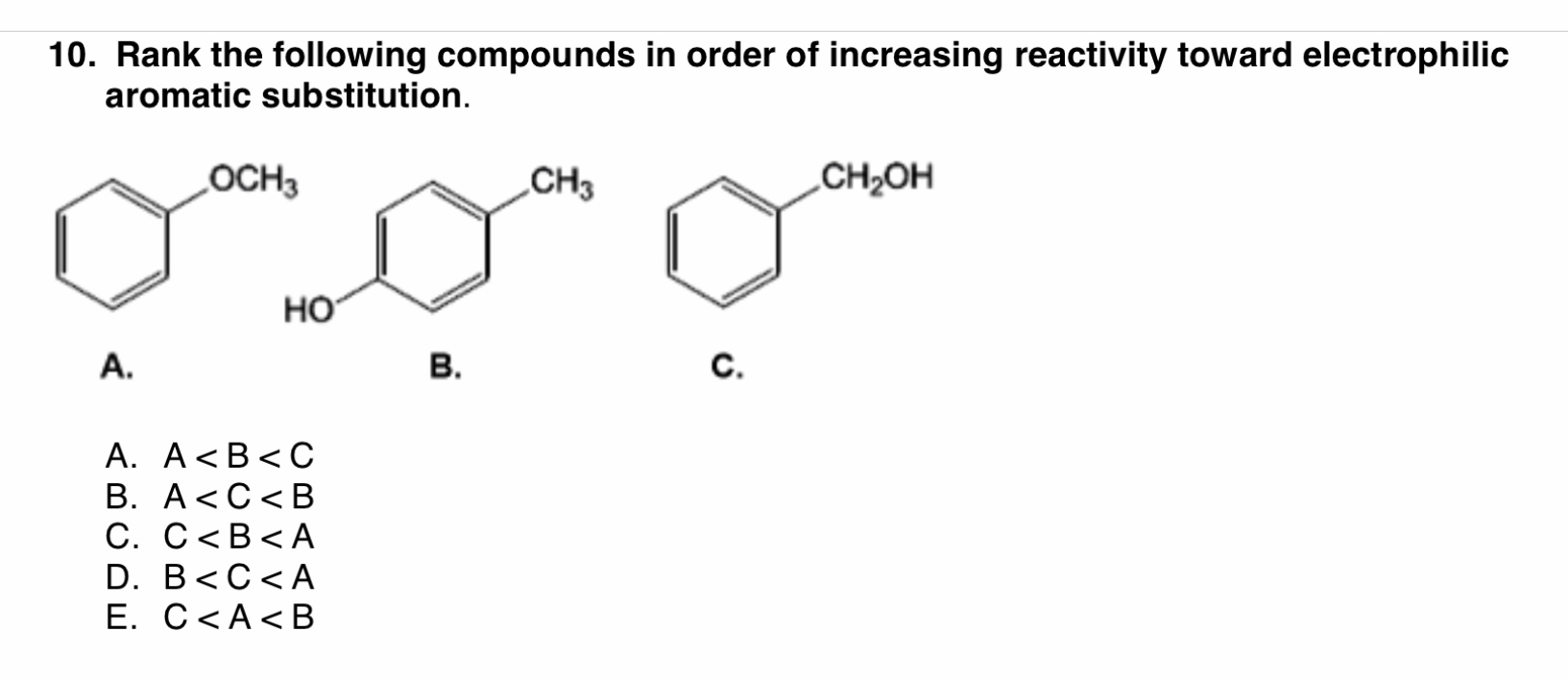
These reactions allow the introduction of different functional groups onto aromatic rings. Electrophilic aromatic substitutions are reactions in which an electrophile replaces one of the aromatic hydrogens. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
